What is element?

What is element? Silver Chemical & Physical Properties
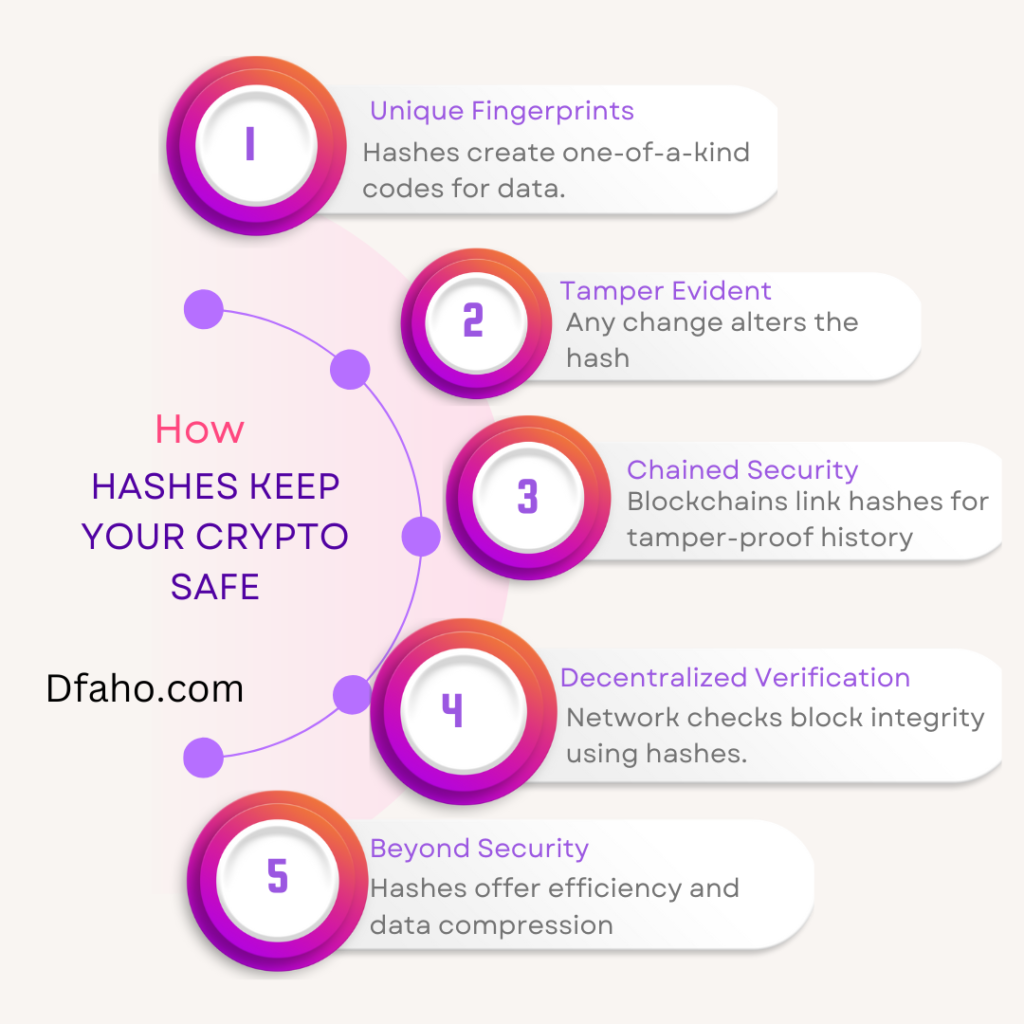
Silver could be a transition metal with part image and number 47. The part is found in jeweler and currency for its beauty and price and in natural philosophy for its high conduction and plasticity.
What is element? Silver Basic Facts
Atomic Number: forty seven
Symbol: Ag
Atomic Weight: 107.8682
Discovery: famous since prehistoric time. Electron Configuration: 5s14d10
Word Origin: Anglo-Saxon Seafloor or sulfur; that means ‘silver’, and Latin argentum that means ‘silver’
Properties: The freezing point of silver is 961.93°C, boiling purpose is 2212°C, and relative density is ten.50, with a valence of one or two. Pure silver includes a good white metallic luster. Silver is slightly tougher than gold. It’s terribly ductile and malleable, exceeded in these properties by gold and atomic number 46. Pure silver has the best electrical and thermal conduction of all metals. Silver possesses rock bottom contact resistance of all metals. Silver is stable in pure air and water, though it tarnishes upon exposure to gas, sulfide, or air containing sulfur.
Uses: The alloys of silver have several business uses. Metal is employed for tableware and jeweler. Silver is employed in photography, dental compounds, solder, brazing, electrical contacts, batteries, mirrors, and written circuits. Freshly deposited silver is that the best famous reflector of actinic ray, however it chop-chop tarnishes and loses its reflectivity. Silver fulminate could be a powerful explosive. Iodide is employed in cloud seeding to supply rain. Chloride will be created clear and is additionally used as cement for glass. Caustic, or silver nitrate, is employed extensively in photography. Though silver it isn’t thought-about cacogenic, most of its salts square measure toxic, thanks to the anions concerned. Exposure to silver shouldn’t exceed zero.01 mg/M3. Silver compounds will be absorbed into the vascular system, with deposition of reduced silver in body tissues. This could lead to agrarian that is characterized by a grayish pigmentation of the skin and secretion membranes. Silver is bactericidal and should be accustomed kill several lower organisms while not damage to higher organisms. Silver is employed as coinage in several countries.
Sources: Silver happens native and in ores including mineral and horn silver. Lead, lead-zinc, copper, copper-nickel, and gold ores square measure alternative principal sources of silver. Business fine silver is a minimum of ninety nine.9% pure. Business purities of ninety nine.999+% square measure offered.
Element Classification: Transition Metal
Silver Physical information
Density: ten.5
What is element? Appearance: silvery, ductile, malleable metal
Isotopes: There square measure thirty eight famous isotopes of silver starting from Ag-93 to Ag-130. Silver has 2 stable isotopes: Ag-107 and Ag-109 .Atomic Radius: a hundred and forty four
Atomic Volume: ten.3
Covalent Radius: 134
Ionic Radius: eighty nine 126
Specific Heat: zero.237
Fusion Heat: eleven.95
Evaporation Heat: 254.1
Debye Temperature (K): 215.00
Pauling Negativity Number: one.93
First ionized Energy: 730.5
Thermal Conductivity: 429 W/make @ three hundred K
Oxidation States: +1, +2, +3
Lattice Structure: Face-Centered cubical
Lattice Constant: four.090
CAS registers Number: 7440-22-4
Silver Trivia:
- Silver’s part image atomic number 47, is from the Latin word argentum that means silver.
- In several cultures, and a few pseudoscience texts, silver was related to the Moon whereas gold was related to the Sun.
- Silver has the best electrical conduction of all metals.
- Silver has the best thermal conduction of all metals.
- Silver salt crystals darken once exposed to lightweight. This method was very important to photography.
- Silver is taken into account one among the noble metals.
- Silver is slightly tougher than gold.
- Silver ions and silver compounds square measure cacogenic to several kinds of microorganism, protectant and fungi. Silver coins accustomed are hold on in containers of water and wine to stop spoiling.
- Silver nitrate has been accustomed stop infection in burns and alternative wounds.
More Silver Facts
Sources
- Ensley, John. Nature’s building blocks: associate A-Z Guide to the weather. University Press. pp. 492–98. ISBN 978-0-19-960563-7.
- Greenwood, Norman N.; Earns haw, Alan. Chemistry of the weather. Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- Hammond, C. R . . . the weather, in reference of Chemistry and Physics. CRC press. ISBN 978-0-8493-0485-9.
- West, Henry Martin Robert. CRC, reference of Chemistry and Physics. Boca Raton, Florida: Chemical Rubber Company business enterprise. pp. E110. ISBN 0-8493-0464-4.